 The molecular chaperone Hsp90 (90 kDa heat-shock protein) is required for managing conformational development, stability and function of proteins in the cellular environment –. 
The funder had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.Ĭompeting interests: The authors have declared that no competing interests exist. No additional external funding was received for this study. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.įunding: This work is partly supported by funding from The University of Kansas. Received: DecemAccepted: ApPublished: May 18, 2012Ĭopyright: © 2012 Dixit, Verkhivker. PLoS ONE 7(5):Įditor: Suzannah Rutherford, Fred Hutchinson Cancer Research Center, United States of America The obtained insights may be useful in guiding discovery of allosteric Hsp90 inhibitors targeting protein interfaces with co-chaperones and protein binding clients.Ĭitation: Dixit A, Verkhivker GM (2012) Probing Molecular Mechanisms of the Hsp90 Chaperone: Biophysical Modeling Identifies Key Regulators of Functional Dynamics. Hence, allosteric signaling through a small network of distantly connected residue clusters may be a rather general functional requirement encoded across molecular chaperones. This mechanistic model of Hsp90 dynamics and function is consistent with the notion that allosteric networks orchestrating cooperative protein motions can be formed by evolutionary conserved and sparsely connected residue clusters. At the same time, the thermodynamics-based “conformational selection” of functional states is likely to be activated based on the nature of the binding partner. The interaction networks regulating functional motions of Hsp90 may be determined by the inherent structural architecture of the molecular chaperone. These findings have provided support to a long-standing assertion that allosteric regulation and catalysis may have emerged via common evolutionary routes. We have found that these functional motifs may be utilized by the molecular chaperone machinery to act collectively as central regulators of Hsp90 dynamics and activity, including the inter-domain communications, control of ATP hydrolysis, and protein client binding. This approach has identified a network of conserved regions common to the Hsp90 chaperones that could play a universal role in coordinating functional dynamics, principal collective motions and allosteric signaling of Hsp90. We combined molecular dynamics simulations, principal component analysis, the energy landscape model and structure-functional analysis of Hsp90 regulatory interactions to systematically investigate functional dynamics of the molecular chaperone. 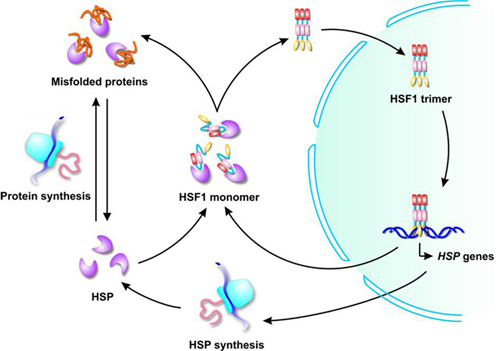
Despite significant advances in understanding structure and function of molecular chaperones, organizing molecular principles that control the relationship between conformational diversity and functional mechanisms of the Hsp90 activity lack a sufficient quantitative characterization. In this review, these structural insights are discussed to give a picture of the current understanding of how Hsp70 chaperones work.Deciphering functional mechanisms of the Hsp90 chaperone machinery is an important objective in cancer biology aiming to facilitate discovery of targeted anti-cancer therapies. In the past few years, structural insights into the molecular mechanism of this allosterically regulated binding have emerged and provided deep insight into the deceptively simple Hsp70 molecular machine that is so widely harnessed by nature for diverse cellular functions. The key to Hsp70 functions is that their substrate binding is transient and allosterically cycles in a nucleotide-dependent fashion between high- and low-affinity states. Central players in protein homeostasis, these jacks-of-many-trades are utilized in a variety of ways because of their ability to bind with selective promiscuity to regions of their client proteins that are exposed when the client is unfolded, either fully or partially, or visits a conformational state that exposes the binding region in a regulated manner. 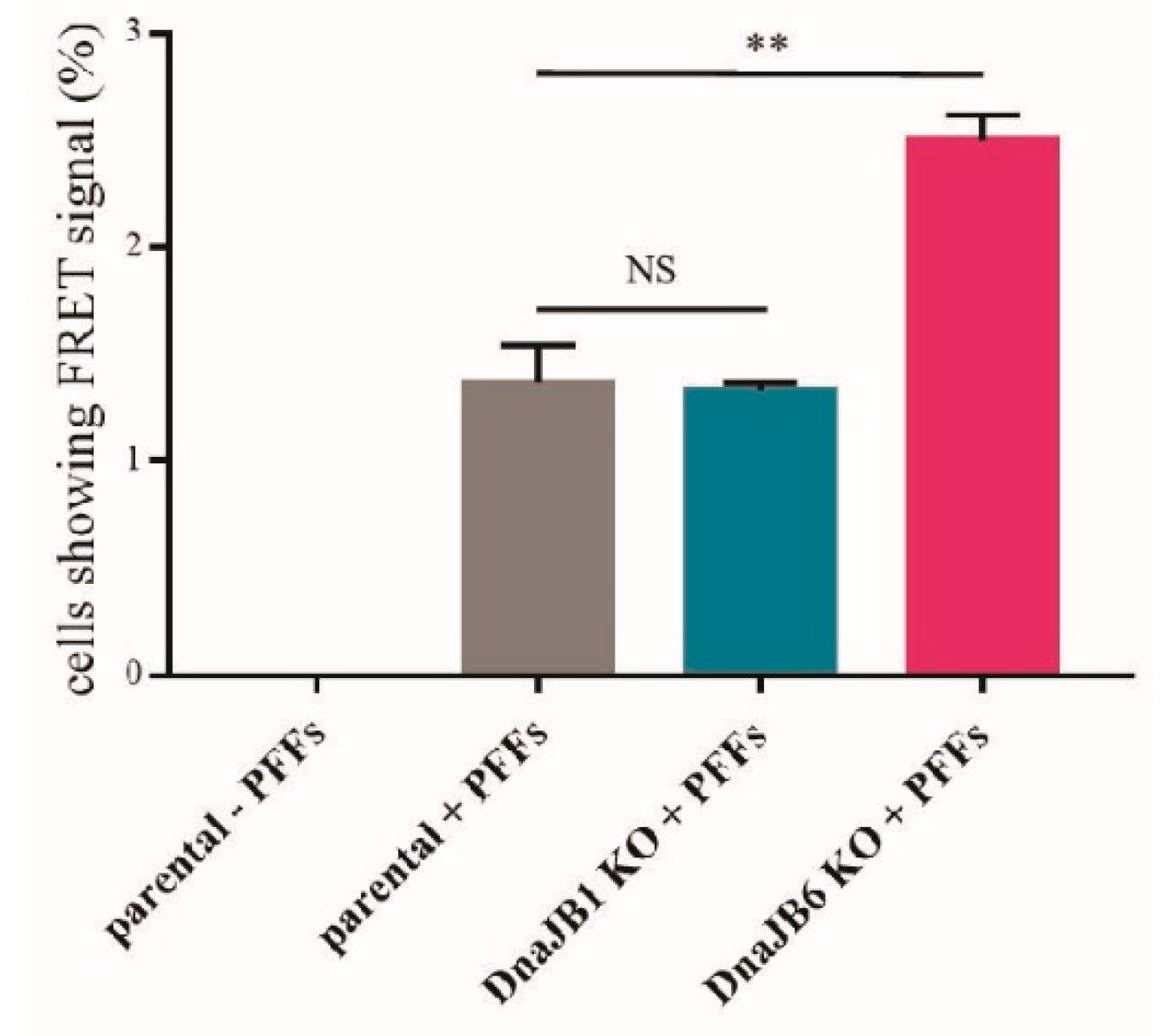
The Hsp70 family of chaperones works with its co-chaperones, the nucleotide exchange factors and J-domain proteins, to facilitate a multitude of cellular functions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
